Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sekine, Yurina; Nankawa, Takuya; Hiroi, Kosuke; Oba, Yojiro*; Nagakawa, Yoshiyasu*; Sugita, Tsuyoshi; Shibayama, Yuki; Ikeda-Fukazawa, Tomoko*
Carbohydrate Polymers, 327, p.121538_1 - 121538_11, 2024/03
Times Cited Count:0 Percentile:0.01(Chemistry, Applied)We describe non-toxic, tough nanocellulose (NC) hydrogels formed from chemically unmodified NC by cellulose crystalline transformation and subsequent freeze cross-linking reaction. Using low-concentration NaOH and freezing together induced the crystalline transformation of NC from cellulose I to II via freeze concentration. After the crystalline transformation, cross-linking between the NC and CA in the freeze concentration layer (FCL) provided a strong NC network structure, forming NC hydrogels with high mechanical strength. The freeze-cross-linked NC hydrogel easily retained powder adsorbents in its inner space by mixing the NC-NaOH sol and the powder, and the hydrogel showed high removal efficiency for heavy metals. The results highlight the versatility of chemically unmodified celluloses in developing functional materials, suggest possible practical applications.
Sekine, Yurina
Cellulose Communications, 30(4), p.203 - 208, 2023/12
In this paper, we introduce the fabrication and properties of carboxymethyl cellulose nanofiber (CMCF) hydrogels with high compression recoverability, macroporosity, and nontoxic properties by utilizing the phase separation phenomenon of ice crystals and solutes that occurs during freezing of aqueous solutions. The features of the developed freeze cross-linked CMCF hydrogel include that it can be made from harmless raw materials with simple procedure, high water content, and biodegradability. By taking advantage of these properties, this hydrogel synthesis technique can contribute to expanding the use of cellulose derivatives as environmental purifiers, functional materials, etc.
Sekine, Yurina; Nankawa, Takuya
Bulletin of the Chemical Society of Japan, 96(10), p.1150 - 1155, 2023/10
Times Cited Count:0 Percentile:0.01(Chemistry, Multidisciplinary)The phase separation of ice crystals and solutes and bound water that occurs during freezing can be used as a reaction field to control a hierarchical structure of hydrogels. Here, we present a study of carboxymethyl cellulose nanofiber (CMCF) hydrogels formed using the solid-quasi liquid phase separation. CMCF hydrogels were formed simply by adding citric acid to frozen CMCF and thawing the mixture. It was found that rearrangement of CMCF structures via hydrogen bonding proceeds in the freeze concentration layer before the ice crystals melt. Under freeze concentration, CMCF and bound water are confined at high concentrations. The crosslinking reaction in such a unique space contributed to the formation of CMCF hydrogel with high mechanical strength. We discuss the gelation behavior and properties of freeze crosslinked CMCF hydrogels and their applications.
Watanabe, Teppei*; Sekine, Yurina; Ikeda-Fukazawa, Tomoko*
Macromolecules, 56(16), p.6217 - 6221, 2023/08
Times Cited Count:1 Percentile:51.7(Polymer Science)To investigate the ability of hydrogels to separate hydrogen isotopes in water, we analyzed the Raman spectra of poly-N,N-dimethylacrylamide (PDMAA) hydrogels containing deuterated water during dehydration. The results show a significant fractionation of hydrogen isotopes during dehydration. The D molar ratio of the hydrogel increases from 0.056 to  0.2 during dehydration from 90.5 wt% to 5 wt% in water content. Deuterated water preferentially forms hydrogen bonds with hydrophilic groups of the polymer in hydrogels because of the difference in strengths of hydrogen bonds between protium and deuterium. As a result, normal water preferentially evaporates in the initial stage of dehydration, leaving deuterated water in the drying hydrogel. The results suggest that hydrogels are an efficient material for isotope fractionation with evaporation.
0.2 during dehydration from 90.5 wt% to 5 wt% in water content. Deuterated water preferentially forms hydrogen bonds with hydrophilic groups of the polymer in hydrogels because of the difference in strengths of hydrogen bonds between protium and deuterium. As a result, normal water preferentially evaporates in the initial stage of dehydration, leaving deuterated water in the drying hydrogel. The results suggest that hydrogels are an efficient material for isotope fractionation with evaporation.
Sekine, Yurina; Kobayashi, Riki*; Chi, S.*; Fernandez-Baca, J. A.*; Suzuya, Kentaro; Fujisaki, Fumika*; Ikeda, Kazutaka*; Otomo, Toshiya*; Fukazawa, Tomoko*; Yamauchi, Hiroki; et al.
JPS Conference Proceedings (Internet), 8, p.033009_1 - 033009_6, 2015/09
Neutron diffraction profiles of low-D O-content poly-N,N,-dimethylacrylamide (PDMAA) hydrogels were measured from 100 to 300 K to investigate the structure and properties of water in hydrogels. A broad diffraction observed at temperatures lower than 220 K indicates the existence of amorphous ice in the hydrogels at low temperatures. During the heating process, diffraction peaks from ice crystals appear in the profiles. These peaks indicate that cubic ice and hexagonal ice exist at T
O-content poly-N,N,-dimethylacrylamide (PDMAA) hydrogels were measured from 100 to 300 K to investigate the structure and properties of water in hydrogels. A broad diffraction observed at temperatures lower than 220 K indicates the existence of amorphous ice in the hydrogels at low temperatures. During the heating process, diffraction peaks from ice crystals appear in the profiles. These peaks indicate that cubic ice and hexagonal ice exist at T  220 K. We refined the structure parameters of these ices in the hydrogel, which indicate the existence of low-density ice in the hydrogel.
220 K. We refined the structure parameters of these ices in the hydrogel, which indicate the existence of low-density ice in the hydrogel.
Sekine, Yurina; Fukazawa, Tomoko*; Aizawa, Mamoru*; Kobayashi, Riki*; Chi, S.*; Fernandez-Baca, J. A.*; Yamauchi, Hiroki; Fukazawa, Hiroshi
Journal of Physical Chemistry B, 118(47), p.13453 - 13457, 2014/08
Times Cited Count:2 Percentile:3.74(Chemistry, Physical)Neutron diffraction patterns for deuterated poly- ,
, ,-dimethylacrylamide (PDMAA) hydrogels were measured from 10 to 300 K to investigate the structure and properties of water in the gels. Diffraction peaks observed below 250 K indicate the existence of ice in the hydrogels. Some diffraction peaks from the ice are at lower diffraction angles than those in ordinary hexagonal ice (Ih). These shifts in peaks indicate that the lattice constants of the
,-dimethylacrylamide (PDMAA) hydrogels were measured from 10 to 300 K to investigate the structure and properties of water in the gels. Diffraction peaks observed below 250 K indicate the existence of ice in the hydrogels. Some diffraction peaks from the ice are at lower diffraction angles than those in ordinary hexagonal ice (Ih). These shifts in peaks indicate that the lattice constants of the 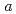 and
and 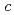 axes in the ice are about 0.29% and 0.3% higher than those in ice Ih, respectively. The results show that bulk low-density ice can exist in PDMAA hydrogels. The distortions in the lattice structure of ice imply significant interactions between water molecules and the surrounding polymer chains, which play an important role in the chemical and mechanical properties of the hydrogel.
axes in the ice are about 0.29% and 0.3% higher than those in ice Ih, respectively. The results show that bulk low-density ice can exist in PDMAA hydrogels. The distortions in the lattice structure of ice imply significant interactions between water molecules and the surrounding polymer chains, which play an important role in the chemical and mechanical properties of the hydrogel.
Wasikiewicz, J. M.; Nagasawa, Naotsugu; Tamada, Masao; Mitomo, Hiroshi*; Yoshii, Fumio
Nuclear Instruments and Methods in Physics Research B, 236(1-4), p.617 - 623, 2005/07
Times Cited Count:54 Percentile:95.02(Instruments & Instrumentation)The absorption ability of various metal ions into EB - radiation crosslinked carboxymethylchitin and carboxymethylchitosan has been investigated. The highest adsorption of Scandium and Gold has been obtained for carboxymethylchitin (CMCht) and carboxymethylchitosan (CMChts), respectively. Kinetic studies showed that adsorption of most of the metal ions occur in a relatively short period of time (2 hours). Detail investigation of adsorption of gold ions has been carried out for both hydrogels. The maximum uptake of Au cations, based on Langmuir equation was determined to be 37.59 mg/g for CMChts and 11.86 mg/g for CMCht. Both hydrogels indicate favorable adsorption of gold cations.
Yagi, Toshiaki
Dai-14-Kai Hoshasen Riyo Gijutsu Semina; Hirogaru Hoshasen No Sangyo Riyo Koen Tekisuto, p.67 - 72, 2004/10
no abstracts in English
Tamada, Masao; Seko, Noriaki; Yoshii, Fumio
Radiation Physics and Chemistry, 71(1-2), p.223 - 227, 2004/09
Times Cited Count:71 Percentile:96.47(Chemistry, Physical)Graft polymerization and crosslinking in radiation processing were attractive techniques to modify conventional polymers. In the case of graftpolymerization, metal adsorbent was prepared by introducing the chelate function to trunk polymer like polyethylene. The obtained metal adsorbent was applied to the recovery of uranium in seawater. Three years' marine experiment revealed that 1kg of uranium could be collected by using graft-adsorbent. Natural polymers such as derivatives of starch and cellulose led irradiation crosslinking at paste-like condition and formed hydrogels. These hydrogels had degradability even after crosslinking. The mat of degradable hydrogel was effective to prevent bedsores as a healthcare product. After use, the hydrogel can be converted to fertilizer by decompositions.
Zhao, L.; Mitomo, Hiroshi*; Yoshii, Fumio; Kume, Tamikazu
Journal of Applied Polymer Science, 91(1), p.556 - 562, 2004/01
Times Cited Count:20 Percentile:54.08(Polymer Science)no abstracts in English
Yoshii, Fumio
Kobunshi Geru No Saishin Doko, p.150 - 163, 2004/00
no abstracts in English
Zhao, L.; Mitomo, Hiroshi*; Zhai, M.*; Yoshii, Fumio; Nagasawa, Naotsugu; Kume, Tamikazu
Carbohydrate Polymers, 53(4), p.439 - 446, 2003/09
Times Cited Count:203 Percentile:99.05(Chemistry, Applied)no abstracts in English
 CO
CO aqueous solutions with electron-beam irradiation
aqueous solutions with electron-beam irradiationXu, L.*; Nagasawa, Naotsugu; Yoshii, Fumio; Kume, Tamikazu
Journal of Applied Polymer Science, 89(8), p.2123 - 2130, 2003/08
Times Cited Count:8 Percentile:32(Polymer Science)no abstracts in English
Zhao, L.; Mitomo, Hiroshi*; Nagasawa, Naotsugu; Yoshii, Fumio; Kume, Tamikazu
Carbohydrate Polymers, 51(2), p.169 - 175, 2003/01
Times Cited Count:115 Percentile:96.32(Chemistry, Applied)no abstracts in English
Wach, R. A.*; Mitomo, Hiroshi*; Yoshii, Fumio; Kume, Tamikazu
Macromolecular Materials and Engineering, 287(4), p.285 - 295, 2002/04
Times Cited Count:58 Percentile:85.14(Materials Science, Multidisciplinary)no abstracts in English
Kume, Tamikazu; Nagasawa, Naotsugu*; Yoshii, Fumio
Radiation Physics and Chemistry, 63(3-6), p.625 - 627, 2002/03
Times Cited Count:95 Percentile:97.79(Chemistry, Physical)no abstracts in English
Yoshii, Fumio
Mizu, (612), p.16 - 22, 2001/06
no abstracts in English
Hendri, J.*; Hiroki, Akihiro*; Maekawa, Yasunari; Yoshida, Masaru; Katakai, Ryoichi*
Radiation Physics and Chemistry, 61(2), p.155 - 161, 2001/05
Times Cited Count:2 Percentile:19.66(Chemistry, Physical)no abstracts in English
Fei, B.*; Wach, R. A.*; Mitomo, Hiroshi*; Yoshii, Fumio; Kume, Tamikazu
Journal of Applied Polymer Science, 78(2), p.278 - 283, 2000/10
Times Cited Count:192 Percentile:98.21(Polymer Science)no abstracts in English
Yoshii, Fumio
Hoshasen To Sangyo, (87), p.55 - 58, 2000/09
no abstracts in English